That autism is a disorder of early brain development is no longer a matter of debate. In particular, studies point to errors in the way young neurons are born, migrate and make crucial connections.
By Jill Escher
Neurodiversity advocates often speak of autism as a sort of benign cognitive difference, but let’s be clear: research does not support this romanticized view. To the contrary, studies increasingly reveal how autism is rooted in abnormal brain physiology and function, and in particular, critical errors in the way the brain wires up during early development.
Autism, at least the disabling autism affecting my children and others like them, is, quite frankly, a birth defect. Just one that’s hidden in mini-structural patterns beneath the skull, and not detectable at birth.
This article offers an overview of what research is teaching us about faulty brain-building and functioning in the genesis of autism, leading to characteristic impairments in thinking, executive function, sensory processing, behavior and social communication. While without question much more awaits to be discovered, neuroscience has advanced to the point where many biological foundations of autism have come into focus and should inform how we understand this increasingly prevalent, and often profoundly disabling, disorder.
HOW THE BRAIN IS MADE
It is helpful to start with a basic understanding of how a human brain, a 3-pound fatty blob of unfathomable complexity, is made. The process starts in the embryo, soon after fertilization. At this stage, the nervous system consists of a hollow tube running along the top of the embryo. The inner surface of this tube is lined with neural stem cells that divide to generate vast numbers of immature neurons, which then migrate en masse towards the outer surface. This happens at different rates (try to imagine a quarter of a million every minute!) along the length of the tube, so that one end forms the brain, and the other forms the spinal cord.
At the front end of the tube, the different regions of the brain are formed, and immature neurons migrate in successive waves to form the six layers of the cerebral cortex — the outer “shell” of the brain, and the part most strongly implicated in autism. To form the cortex each subsequent wave of young neurons migrates past the one before it, a process guided by cells called glia. This process of neuronal generation and migration is largely complete by 5-6 months of gestation.
Schematic of neural migration to form the cortex in the early brain. Glia cells are depicted in green. Neurons (blue) are born in the ventricular zone (VZ) and migrate along the glia fibers. The cortex is formed in an inside-out fashion such that later-born neurons pass through earlier-born neurons in deeper layers (lighter blue shading). In autism, the complex process of neural migration, placement, alignment and connection is impaired.(Diagram from Gilbert and Man, Front. Cell. Neurosci., 2017)
Once a young neuron has reached its final destination, it sprouts extensions called axons and dendrites that extend towards, then begin to form connections with, other neurons. Dendrites from just one neuron can have thousands of spines, with each spine forming a synaptic connection upon maturing. The number of spines and connections are fine-tuned in early development.
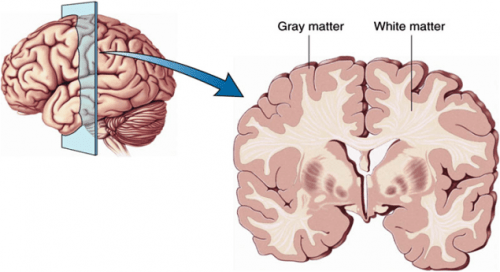
Diagram of grey and white matter of the cortex.
Though just a few millimeters thick, the cortex comprises six layers, and in normal brains, the cells are arranged uniformly in each layer, like well connected microprocessors. Complex thought and behaviors depend on precise organization and robust connectivity of these cortical cells. During months 6-7, the cortex folds, tripling in area, becoming the largest structure in the human brain. Neurons continue the process of connecting with each other both within and across brain regions through the early postnatal period. The sections of neurons within the cortex are known as grey matter, and the deeper tissues, or white matter, is composed of axons extending from the neurons to other parts of the brain.
In all, an estimated 86 billion neurons must proliferate, grow and migrate to the correct locations, and then connect robustly with other neurons, with 100s of trillions of connections. Then even after the organ takes its basic shape, the neurons and their neurotransmitters, which send signals across the synapses, must have the capacity to function properly as well.
In addition, neurons are not the only type of brain cells, and certainly not the only brain cells involved in autism. Glial cells, including microglia, astrocytes, and oligodendrocytes constitute a large fraction of the human brain. The microglia, to describe one, are the brain's 'housekeeping' cells, which constantly patrol the organ and migrate to sites of injury and infection, where they engulf cellular debris and microbes. They also gobble up unwanted synaptic connections, a function that is crucial for sculpting developing brain circuits, and for maintaining mature circuits. The size of the brain doubles in the first year of life, not because new neurons are formed, but because of the expanding glial cell network.
ABNORMALITIES IN THE AUTISM BRAIN
So how does neural development deviate from the norm in autism brains? Let’s first acknowledge that one cannot simply biopsy the brains of people with autism to find these answers. Because brain tissues are inaccessible, scientists use indirect techniques, including analysis of post-mortem autism brains, neuroimaging techniques such as functional magnetic resonance imaging (fMRI), in vitro (think petri dish) studies where neurons may be programmed from patients’ other cells such as blood or skin cells, and animal models, for example, looking at brains of mice with certain autism-related mutations.
Studies using various versions of these techniques have found strong and consistent evidence that critical processes of cortical development, including cell birth, migration, and/or synaptogenesis are disrupted, resulting in abnormalities including impaired micro-structural development and failure of proper connectivity. Neurons get stuck in migration, or don’t file into proper position, or fail to sprout or lose connections. The excitatory and inhibitory functions of neurons are unbalanced. Strong evidence also supports the role of synaptic dysfunction. The end result is that multiple stages of neuronal development are disrupted, including those in the cortex, with neurons less able to coordinate their actions with other cells in their surroundings.
Dendritic impairments. A simple schematic showing decreased dendtric spines in a pyramidal neuron in an autism brain. (From Zoghbim Science 3003)
These developmental failures can have profound consequences. For example, the frontal lobe of the cortex performs complex mental functions such as reasoning and decision-making, and also contains the motor areas, which plan and execute voluntary movements. The prefrontal cortex is tied to executive functioning (the process guiding goal-directed actions and the ability to handle novel situations), attention, mental flexibility, problem-solving, verbal reasoning, working memory and the ability to switch back and forth between different tasks. When the cortical neurons are not properly connected, these all-important processes, which are deficient in autism cases, are compromised.
Another important risk is abnormal circuit function arising from improper balance of the activity between excitatory and inhibitory neurons. When projection neurons become overactive where there is not enough inhibition, epileptic seizures can result.
Of course the story does not end there. Macrocephaly, or oversized head, occurs in about 15% of autism cases, a phenomenon seen by 18 months of age, often before the onset of symptoms. Microcephaly, or abnormally small head, is seen in about 20% of cases. The big brain of autism seems to result from a pathological overabundance of neurons resulting from disturbed processes of neurogenesis in utero, followed by aberrant dendrite growth, branching, and pruning into the toddler years. Many other brain structures and processes are also implicated in autism, including the amygdala, cerebellum, corpus callosum, and left postcentral gyrus, to name some. Just as there are “many autisms” there is diversity in the brain pathology of those who exhibit autism symptoms.
But if there is a central theme from neuroscience it is that abnormal connectivity patterns — both over-connectivity and under-connectivity — compared with typically developing individuals, precedes the impairments in communication, abstract thought, sensory processing, social attention, repetitive behaviors and learning seen in autism.
BUT WHAT CAUSES BRAIN DEVELOPMENT TO GO AWRY IN AUTISM?
So what causes these mishaps in brain development? These failures do not happen out of the blue; rather, research strongly suggests they are driven by dysregulated expression of the genomic code. While some cases of autism may involve direct insults like certain in utero drug exposures like valproic acid, or perinatal complications like prematurity, by and large the neurodevelopmental pathologies of autism arise due to faulty instructions from the child’s genome. This is not to say that these cases are “genetic” in the classic sense — in fact only a small fraction fit that pattern. But how brain development and function genes are read and put to work is widely seen to deviate from the norm. Most of the autisms may not be genetic per se, but they are innate, rooted in how the genome directs the form and function of brain cells.
The genome can be thought of as an instruction book for how and when to synthesize proteins and differentiate to the end product, be it a brain cell, skin cell, or liver cell. The instruction-book has many parts. Protein-coding genes are the 1-2% of DNA that are transcribed into messenger RNA, which are then translated into polypeptide chains to make proteins. Non-coding DNA does not provide instructions for making proteins, but some of it helps control gene activity, determining when and where genes are turned on and off. Chromatin is the name for the overall physical structure of the chromosome itself, including its protein scaffolding. It helps control what the DNA does—including what DNA is accessible for protein synthesis. The epigenome includes many molecules that attach to DNA and its structure, or influence how genes are transcribed into proteins. All of these elements work together to craft the neurons and determine where and when they are born, migrate and connect up. And all of these processes have been implicated in autism.
Studies of protein-coding, or “exome,” genes have, by a wide margin, received the most attention and funding in autism research. However, this hunt for autism genes has been rather a formidable failure. No “genes for autism” have been found (see Myers et al. AHJG, 2020), and the dozens of exome genes and deletions linked with autism, are, for the most part, causative of multi-dimensional syndromes that most people do not think of as autism: for example, Fragile X, Pitt Hopkins Syndrome, Phelan-McDermid Syndrome, Angelman Syndrome, and Rett syndrome. In some instances the genes are inherited, as with Fragile X, but the majority of the time a mutation arises anew, or “de novo,” in a parent’s sperm or egg or in the conceptus, or during the earliest stages of embryonic development (called somatic mosaicism).
Most autism-linked mutations are extremely rare, occurring in far less than 1% of the autism population, and so far, taken together, these DNA sequence errors can be detected in only about 10% of cases. Nevertheless, these genes are instructive: they typically play important roles in brain development —for example, regulation of neurogenesis or synaptogenesis, whereby connections between newborn cells are formed. For example, some autism-related genetic mutations cause defects in stem cell division, leading to an overproduction of immature neurons, which alters the migration patterns of the cells and disturbs the formation of layers in the developing cortex. Others cause defects in the function of microglial cells, leading to a failure to remove unwanted synapses in the developing neural circuits.
In recent years, researchers have also discovered autism-associated mutations in non-coding sections of the genome. For example, deletions in chromosome region Xp22.11 are sometimes found in males with autism — in non-coding regions, reducing activation of the NMDA receptor, a cell-surface protein that is critical for signaling in the brain, and that plays important roles in learning and memory.
However, in the vast majority of cases, autism causes are unknown, and by my reading of the literature, finding the molecular factors disrupting the proper expression of brain development genes stands as the greatest puzzle in autism research today. Since autism is strongly heritable, the factors likely reside somewhere in the germ cells of the parents — but based on extensive evidence over two decades of research, likely not within the DNA sequence.
But wherever the faults may lie, somehow gene networks highly expressed during fetal brain development are indeed perturbed, impacting crucial signaling pathways for brain-building. This was most recently seen in a study of neurons cultured from blood cells of young children with autism, finding consistent dysregulation in a number of neural pathways. Further, the degree of dysregulation of gene expression correlated with the severity of ASD symptoms in children. Additionally, post-mortem studies have also found striking changes in gene expression in autism brains. For example, a 2016 study examining 48 post-mortem autism brains revealed that transcription levels of 584 genes were elevated in the brains of the autistic individuals compared to those of controls, whereas the levels of another 558 were reduced. Many of the dysregulated genes are expressed in microglia or neurons in the cerebral cortex. Another recent study detected almost 700 differences in gene activity in samples from 15 post mortem autism brains. The biggest differences were found in genes expressed in cells called inhibitory interneurons, which control local circuit activity, in 'projection' neurons in the upper layers of the cortex, which send fibers to neighboring regions of the brain, and in microglial cells. Dysregulation of specific sets of genes in projection neurons and microglia was associated with the severity of autism symptoms.
SUMMING UP
“In sum, while generally speaking autism is not a genetic disorder, it is a disorder of regulation of genes critical for brain development.”
In sum, while generally speaking autism is not a genetic disorder, it is a disorder of regulation of genes critical for brain development. The dysregulation disrupts successive stages of development, including cell birth, migration, dendritic growth, synapse maturation, and the assembly of circuits in the developing cerebral cortex, ultimately leading to the cognitive and behavioral dysfunctions we label as autism. Atypical gene activity is linked to the degree of autistic impairments, with those who are most impaired showing the most atypical activity.
Why does this matter? Understanding the true pathophysiology of autism should have major implications across research, medicine, and social services. For example, knowing the forces that disturb the key gene networks could lead to measures for prevention. Instead of throwing darts at costly and ineffective medical and behavioral treatments, we could more precisely and meaningfully target intervention. We could stop hitting our heads against the wall with nonsense causation theories. But most importantly, by knowing that behavior is the outgrowth of biological short-circuits (which were no one’s fault!) we could more fully understand and support all those who are disabled by autism.
And now, a plea. Autism BrainNet accelerates brain research by fostering the collection and sharing of the rare resource of human brain tissue. Please sign up here to be a participant in the Autism BrainNet Registry. You will receive information on the value of making a brain donation to support research on autism. This is not a consent form and you are under no obligation to ever make a donation. Thank you.
Jill Escher is the president of the National Council on Severe Autism, an autism research philanthropist (Escher Fund for Autism), and the mother of two children with nonverbal forms of autism. With thanks to neuroscientist and writer Moheb Costandi for providing helpful background information.